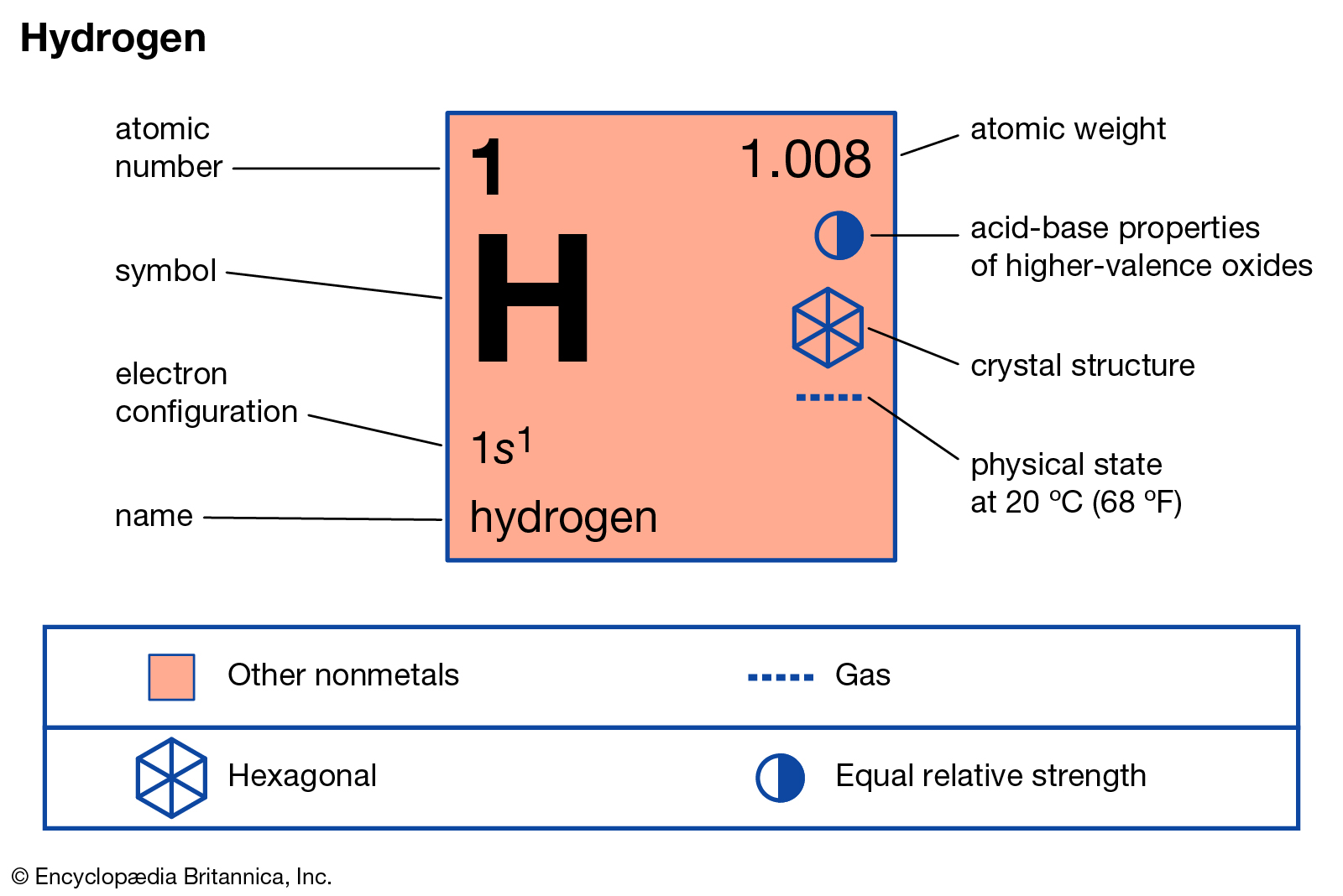
Since 1953, the steady strong substance lithium deuteride (\) has been utilized instead of both deuterium and tritium. The atomic combination of deuterium iotas or of deuterium and the heavier hydrogen isotope, tritium, at high temperature is joined by arrival of a huge measure of energy such responses have been utilized in nuclear weapons. "The Ame2016 atomic mass evaluation (II).Hint: Deuterium, (\ or \ substantial water) is made sure about when the measure of water has been decreased to around 100 thousandth of its unique volume by proceeded with electrolysis. Deuteron, with an atomic weight of 2.014, is the principal. ↑ M.Wang G.Audi F.G.Kondev W.J.Huang S.Naimi X.Xu (2017). Deuterium is present in the oceans in an amount of approximately 156.25 parts per million.This isotope is a stable atomic species that can be found in natural hydrogen. The atomic weight of this component is about 2.014 making it one of the heavier isotopes of hydrogen. The mass number A of each nuclide is an integral sum of protons Z and neutrons N in the nucleus, while the nuclide mass. The nucleus of the deuterium is commonly regarded as deuton or deuteron which is denoted by the chemical symbol of 2H or D. Heavy water contains deuterium atoms and is used in nuclear reactors. The hydrogen in normal water consists of about 99.98 (by weight) of normal hydrogen ( 1 H). Heavy water consists of water molecules with two deuterium atoms instead of the two normal hydrogen atoms. The mass of an electron (0.000549 unified atomic mass units u) is negligible compared to that of protons (1.007276 u) and neutrons (1.008665 u). Deuterium atoms have about twice the atomic mass of normal hydrogen atoms as a result. Generally the value of a tracer arises from the fact that, although its difference in mass or its radioactivity permits its detection, it is essentially active in the same way that the ordinary atoms of the element are. ↑ Thomas Jefferson National Accelerator Facility - Office of Science Education (2010)It's Elemental - The Element Hydrogen. Binding energy per nucleon of each nuclide as a function of the mass number A. Both deuterium and tritium are useful as isotopic tracers for the investigation of chemical structures and of reaction mechanisms.Deuterium is used widely as a tracer for analyzing chemical. Mass of deuteron is expressed in terms of an atomic mass unit (amu) or electron volts (eV). An isotope of hydrogen whose nucleus has one proton and one neutron and whose atomic mass is 2. It is denoted by D or 2H and is called Hydrogen-2. It naturaly occurs in nature because of interaction of cosmic rays with gases found in the atmosphere. Deuterium is a stable atomic particle containing a proton and neutron. It is a radioactive atom which has the half-life of 12.32 years. Its nucleus contains one proton and two neutrons. The atomic number of deuterium is 1 and the mass number is 2. It is the second most common isotope of hydrogen found in nature but it makes up to only 0.0026 – 0.0184% of all atoms.ģH (tritium) has an atomic mass of 3.01604928199(23) u. Carbon atoms with 7 neutrons have an atomic mass of 13 (6 protons + 7 neutrons 13). If one adds a neutron to the nucleus, the atom is still hydrogen, but the atom will have a different atomic weight. To get a more precise figure, we will use the atomic masses from Appendix A. Deuterium has an atomic mass of about 2 and tritium has an atomic mass of about 3, for a total of about 5 g per mole of reactants or about 200 mol in 1.00 kg. It is another stable atom of hydrogen which core has one proton and one neutron. To find the total energy released, we must find the number of deuterium and tritium atoms in a kilogram. It is the most common isotope of hydrogen which makes up to 99.98% of all hidrogen atoms found in nature.ĢH (deuterium) has an atomic mass of 2.01410177811(12) u. Protium has an atomic weight of 1, while deuterium has an atomic mass of 2, and tritium has an atomic mass of 3. Its core consists of only one proton and does not have any neutrons. One of the hydrogen's most exciting features is that it exists in the form of 3 isotopes, different in their atomic composition: protium, deuterium and tritium. Hydrogen is the most abundant element in the universe with a proton and electron number of one. Thus deuterium accounts for approximately 0.0156 (0.0312 by mass) of all the naturally occurring hydrogen in the oceans. How common is deuterium Deuterium has a natural abundance in Earth’s oceans of about one atom in 6420 of hydrogen. The atomic mass can be given as 2.014 amu. It has an atomic number of 1 and an atomic weight of 1.00794. The atomic number of deuterium is 1 and the mass number is 2. Hydrogen is a gas at room temperature, found in Group 1.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
